Quality
When selecting a manufacturing partner, few traits matter more than an unwavering commitment to quality. We have built our reputation through rigorous quality control across every stage of the production process.
When selecting a manufacturing partner, few traits matter more than an unwavering commitment to quality. We have built our reputation through rigorous quality control across every stage of the production process.
Micron Solutions recognizes that whether you require orthopedic implants, medical devices, or consumer goods, defect-free products are non-negotiable.
Micron Solutions is committed to providing customers with products and services that exceed expectations. We are registered and certified for several industry standards, as seen below. Micron's Quality System is audited and certified by Dekra Global Certification Body.

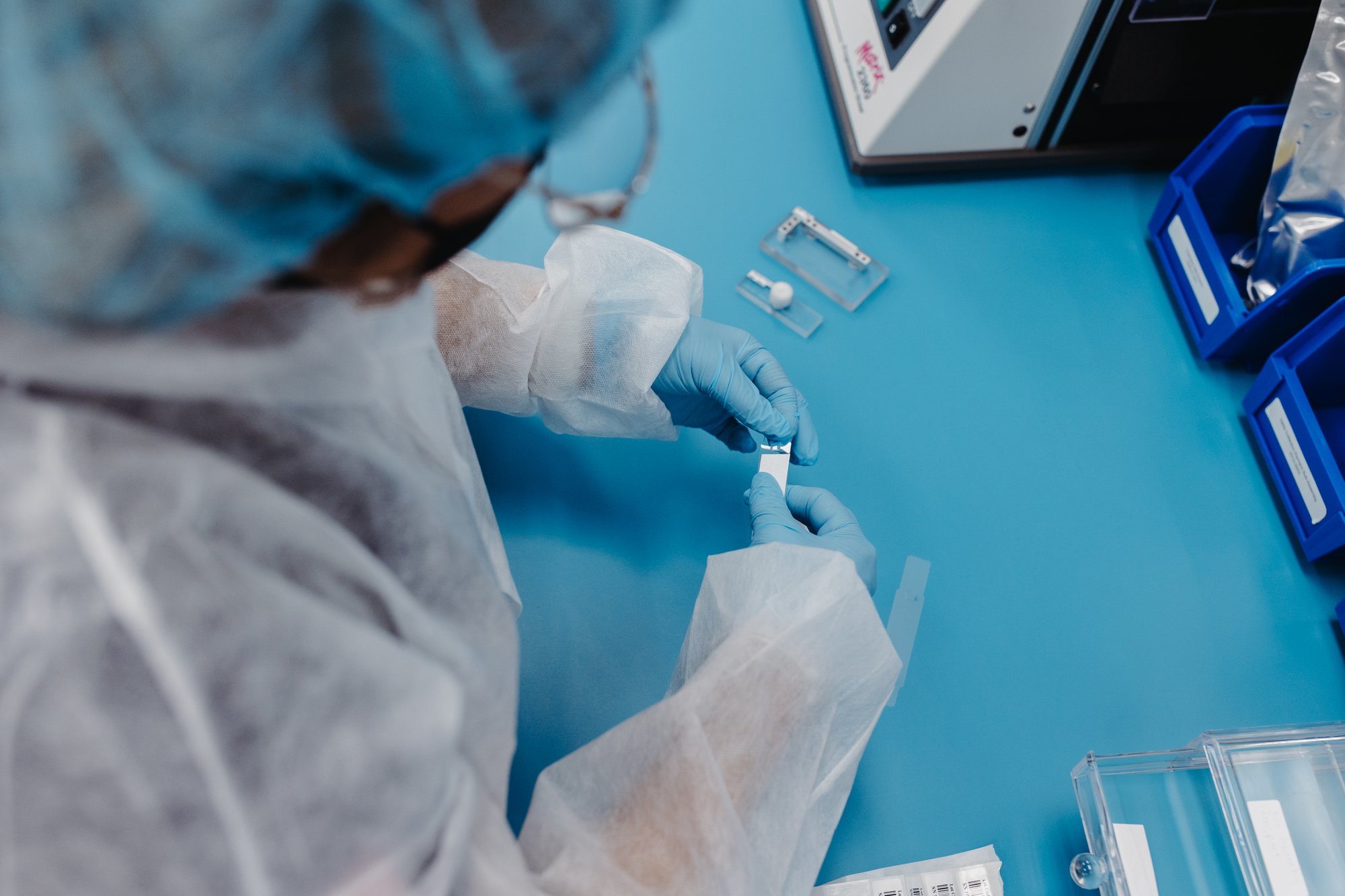
ISO 13485 is the international compliance standard for quality management systems for companies that manufacture medical devices.
Click here to view Micron's ISO Certificate
21CFR part 820 is the U.S. FDA's quality system requirements that includes current good manufacturing practice (cGMP) requirements as set forth in this quality system regulation. The requirements govern the methods used in, and the facilities and controls used for, the design, manufacture, packaging, labeling, storage, installation, and servicing of all finished devices intended for human use.


Copyright © 2023 Micron Products INC. All rights reserved.