For manufacturers, the label represents one of the most consequential steps in the entire production process. In the drug delivery device industry, a labeling error can mean a product recall, a regulatory action, or worse, harm to patients.
The Importance of Getting Labeling Right in Drug Delivery Device Assembly
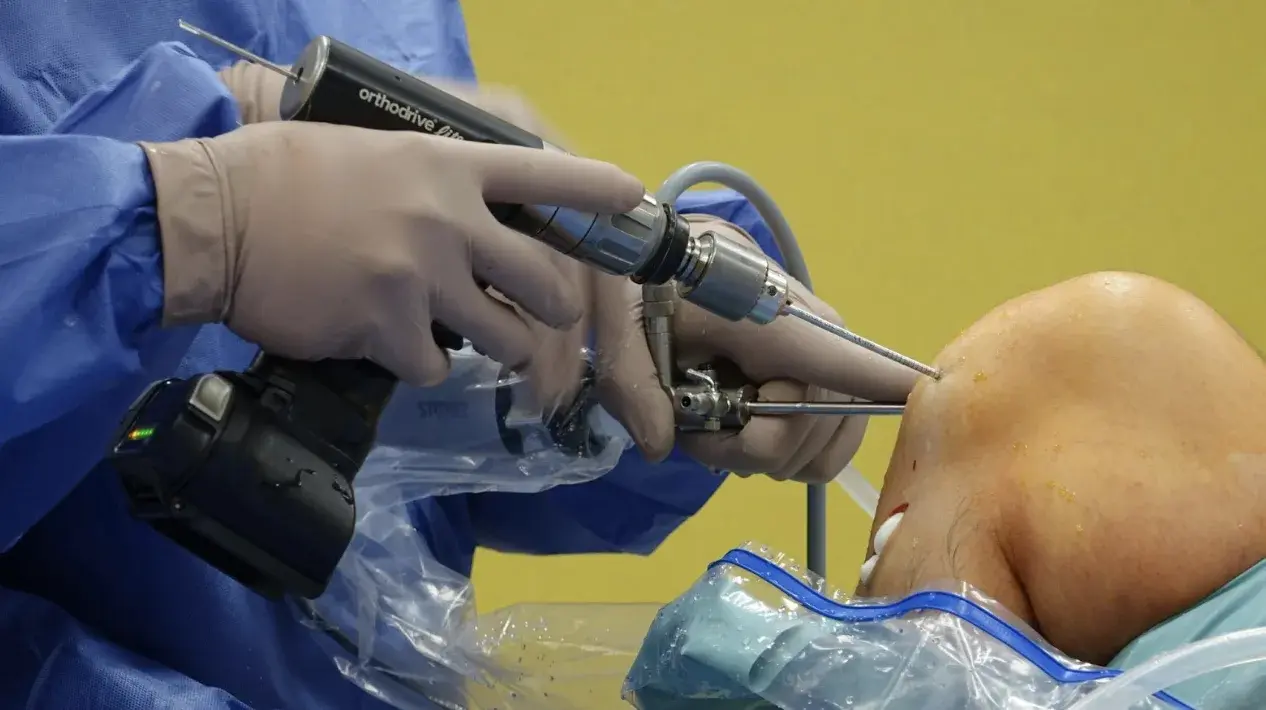
4 Things Orthopedic Surgeons Should Understand Before Developing an Orthopedic Device
Having spent time in the OR, you've seen devices that work brilliantly and others that fall short in ways only a surgeon would notice. So naturally, you're thinking: “I could design something better.”
Micron Solutions Welcomes Dan Hartman to the Team
 We’re excited to welcome Dan Hartman to Micron Solutions as our new Vice President of Sales.
We’re excited to welcome Dan Hartman to Micron Solutions as our new Vice President of Sales.
The Importance of DFM in Medical Device Development
Design for Manufacturability (DFM) in medical device development is the practice of designing products so they can be manufactured easily and cost-effectively. When early design decisions can lock in roughly 80% of a product’s manufacturing costs, involving manufacturing considerations at the design stage is essential. By aligning design with manufacturing realities from day one, medical device teams can avoid expensive changes down the line, reduce quality risks, and accelerate time to market.
Micron Solutions Awarded MMAP Grant to Advance Manufacturing Operations
On October 21st, 2025, Micron Solutions received grant funding from the Massachusetts Manufacturing Accelerate Program (MMAP) during a ceremony at Gillette Stadium. The award represents a major step forward for the company and provides resources to make strategic capital investments that have been in development.
Micron Passes ISO 13485 Audit with No Findings
Micron Solutions has once again passed its ISO 13485 audit with zero findings. This achievement reflects our continued dedication to quality, regulatory excellence, and building trust with our partners in the medical device industry.
Case Study: Blood Collection Device Manufacturing for Clinical Trials and Research
Micron Solutions, a leader in medical device manufacturing, has set new standards in the production of blood collection devices, specifically focusing on finger prick self-collection devices used in clinical trials and research.
3D Printing vs. Injection Molding
In the world of manufacturing, two processes stand out as powerful tools for creating high-quality products: 3D printing and injection molding. Both techniques offer unique advantages and have carved out their respective niches in various industries. As a leading manufacturing company, we understand the importance of choosing the right process for each project, and in this article, we'll delve into the key differences between these two methods.
Case Study: Manufacturing a Drug Delivery Device for Chemotherapy Support
We are happy to support proven innovation making a difference - a drug delivery device designed to start supportive medication on the same day as chemotherapy and deliver the full dose the following day. It’s a smart solution that simplifies treatment timing and improves patient outcomes.
Biocompatibility of Knee Implant Materials
The success of a knee implant depends not only on mechanical performance and design precision but also on the biocompatibility of the materials used.
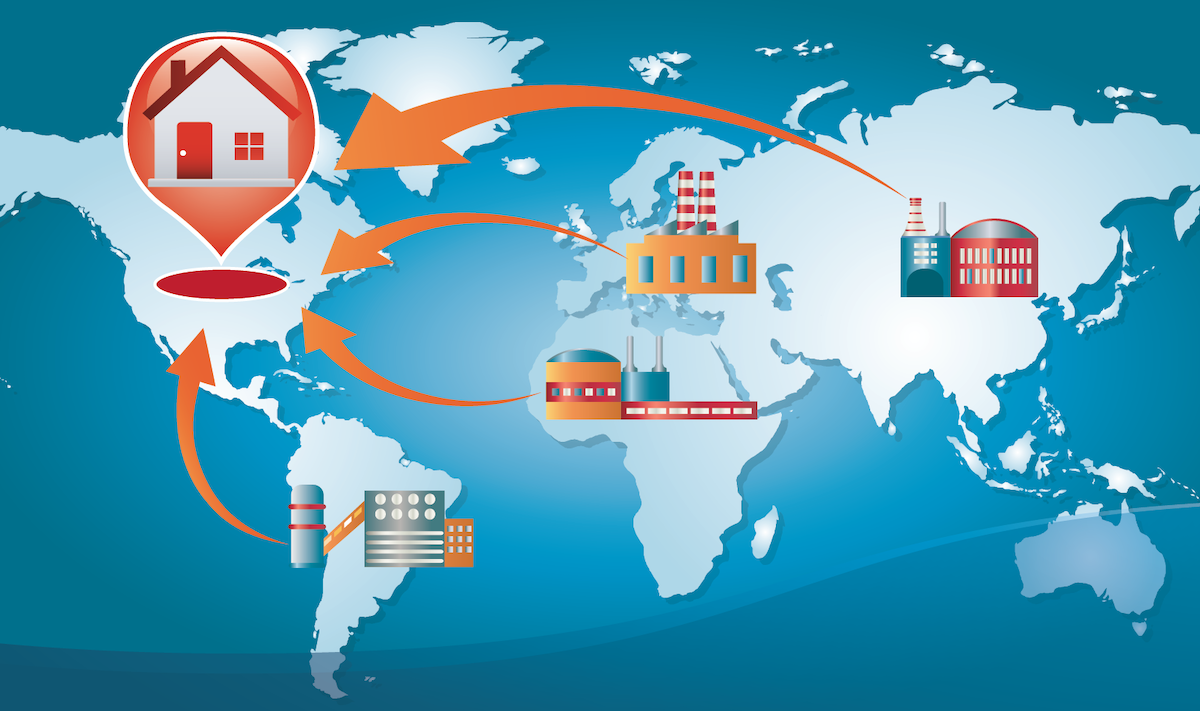
Why USA Manufacturing Matters More Than Ever
Global trade tensions and tariffs have made it more expensive and unpredictable to rely on overseas suppliers. Many U.S. companies have faced unexpected costs; not just in dollars, but in time and customer trust.
Pursuing MA Grants: MCP Cybersecurity Grant, MMAP, and M2I2
Our team is always looking for ways to enhance our capabilities, improve cybersecurity, and expand our manufacturing efforts. Recently, we’ve started exploring grant opportunities that align with our vision for growth and innovation. Three key programs that we are pursuing include: the Massachusetts Cybersecurity Program (MCP) Grant, the Mass Manufacturing Accelerated Program (MMAP), and the Massachusetts Manufacturing Innovation Initiative (M2I2).
Collaborating for a Better Fitchburg
The other week, Micron Solutions had the honor of hosting key leaders from the City of Fitchburg, highlighting the strong and ongoing collaboration between our organization and the city’s leadership. With Micron being one of the top employers in the area, this partnership underscores a shared vision: driving economic growth, creating job opportunities, and building a stronger community in Fitchburg.
Upcoming Events in Spring 2025
This spring, Micron Solutions is excited to attend two major industry events: the Medical Design & Manufacturing (MD&M) West Expo in February and the American Academy of Orthopaedic Surgeons (AAOS) Annual Meeting in March. These events are a great chance for us to meet with partners, industry leaders, and peers to exchange ideas and explore the latest developments in our field.
Micron Solutions at AAHKS 2024 Recap
From November 7–10, 2024, the American Association of Hip and Knee Surgeons (AAHKS) Annual Meeting gathered leading minds in orthopedics in Dallas, Texas. The event is one of the most important gatherings for hip and knee surgeons, offering a platform to discuss implant design, surgical techniques, outcomes, and the challenges shaping modern healthcare.
Automation and Human Collaboration in Drug Delivery Device Manufacturing
At Micron Solutions, a contract manufacturer in Fitchburg, MA, we combine our workforce with our technology (“manomation”) to help scale projects. Our strategy allows us to compete effectively in the market, focusing on scalability, Design for Manufacturing (DFM), Design for Assembly (DFA), and Design for Automated Assembly (DFAA) principles. This whitepaper highlights the importance of early collaboration, cost transparency, and strategic planning in achieving optimal results.
Engineering Precision in Sensor Manufacturing for EEG Applications
The electroencephalogram (EEG) is a neurological test that measures the electrical activity in the brain. At the heart of this technology lie the sensors or electrodes that attach to the scalp, capturing the signals coming from the brain. We at Micron Solutions are proud to be manufacturing a product that makes such a difference in people’s lives.
Case Study in Advanced 2-Shot Molding: Specialty Drug Delivery Devices
As the medical device industry continues to evolve, the demand for innovative and user-friendly medical devices has never been greater. One device that is transforming the lives of patients is a wearable drug delivery device, a critical component that allows individuals to manage their chronic conditions with ease and precision.
Update from February 2024 Trade Events
The Micron team had an enjoyable and memorable time connecting with customers, suppliers, and industry leaders at a MD&M West and AAOS 2024.
Upcoming MedTech Events: Feb 2024
We have two major conferences and expos on our calendar in the coming weeks. If you will be at either of the below events and would like to meet, please reach out! We are always looking to make new connections and strengthen old ones.
Traveling for Customers: India & Malaysia
As a leading contract manufacturer with over 52 years of experience, Micron Solutions provides full-service solutions for medical device innovators across the globe. Having worked with customers across 4 continents, Micron Solutions is a trusted partner to medical technology companies from early prototype development through manufacturing, assembly, packaging, and testing.
Human-Automation Collaboration in Manufacturing
In the evolving landscape of manufacturing and technology, Micron Solutions stands out as a leading company dedicated to innovation and quality. With a distinctive approach of maintaining a 10:1 ratio of people to machines, we have carved a niche for ourselves in the competitive world of manufacturing. This balance between automation and human resources shows how committed we are to achieving the best results possible.
Micron Solutions Returns from MEDICA 2023 with New MedTech Insights
MEDICA 2023 kicked off on November 13th in Düsseldorf, Germany, bringing together thousands of exhibitors and attendees from across the global medical technology landscape. As a leading manufacturer of cutting-edge medical devices, Micron Solutions is thrilled to have participated in this premier industry event.
Medical Design and Manufacturing (MD&M) West 2022 Event
Last week, part of Micron Solution’s upper management team attended the MD&M West convention in Anaheim, California. This event was a huge success with over 13,000 attendees all working toward the collective goal of pushing the boundaries of the medical industry.
Micron Solutions Wins 2021 'Manufacturer of the Year' Award
The team at Micron Solutions has always held themselves to the highest standards in the industry. This is something that we pride ourselves on as a core competency that defines the way in which we work on a day-to-day basis.
American Academy of Orthopaedic Surgeons (AAOS) Annual Meeting 2022
Thank you for joining us at this year’s American Academy of Orthopaedic Surgeons! We had a great time meeting so many new faces and discussing the latest developments in the industry.
We want to take a moment to thank the AAOS for their continued excellence in organizing such an important event.
Events like these allow for great minds to come together and work towards a collective goal of providing orthopaedic surgeons with the best tools and resources, allowing them to continue to provide top-quality care to their patients.
After a great conference, we at Micron Solutions can say that we have never been more proud to work with the AAOS as we strive to build a better world. We hope to see you all at next year’s event!
America COMPETES Act: How it Will Benefit Domestic Manufacturing
In February, the United States passed the ‘America COMPETES Act of 2022’ in the House of Representatives. This act is primarily focused on the development of research and innovation for the manufacturing sector.
Comparing Manufacturability of Orthopedic Implant Materials
Everything in manufacturing is intentional, from the machines utilized to the processes and operations in production— and material is no exception. Medical device producers around the world use a variety of materials, but metals, polymers, and ceramic are the most popular materials for orthopedic implant manufacturing. When manufacturers decide upon what type of implant they want to produce, they first assess its intended purpose and verify that it will meet the patient’s needs. After making this determination, material selection follows suit. The type of implant needed is always directly correlated with the material used.
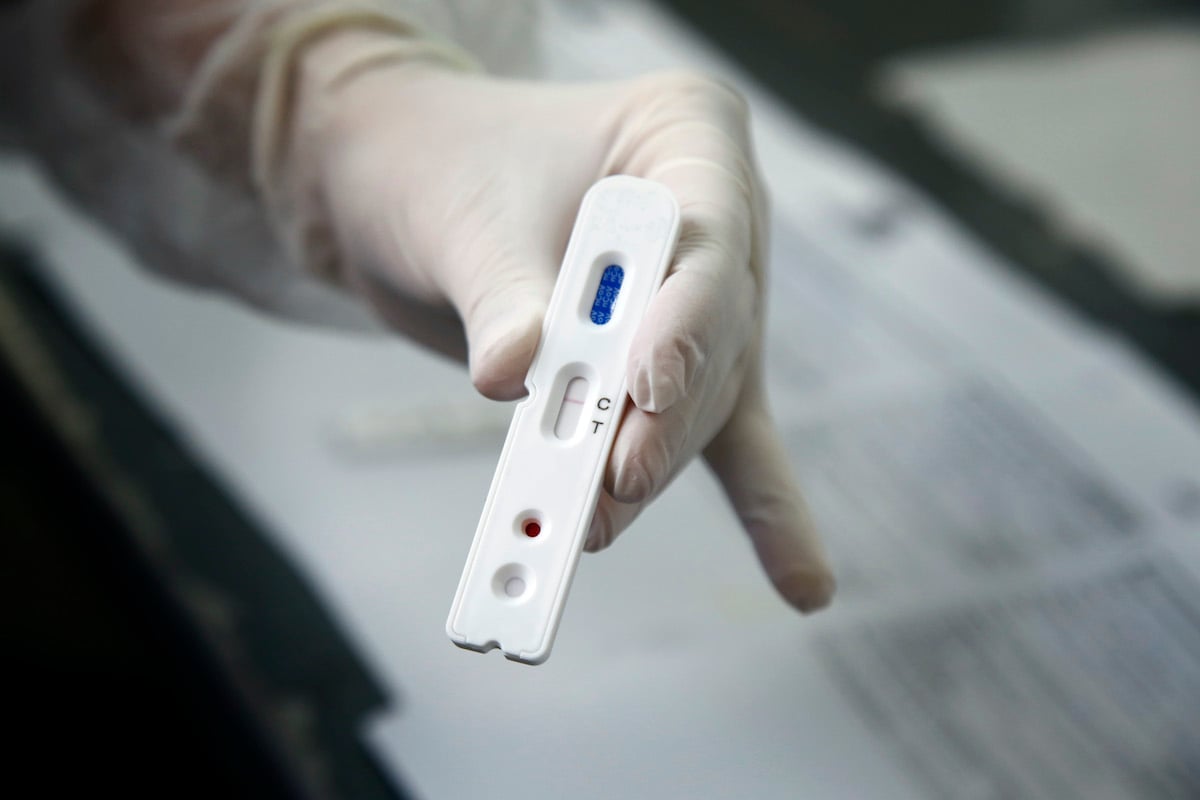
Manufacturing Efficiency Key In Expanding COVID-19 Test Kit Availability
With new strains introducing unique challenges in the fight against coronavirus, the demand for efficient and reliable COVID-19 test kits that can be used in the home has become increasingly amplified. Recently, medical device producers have reached out urgently to leading medical contract manufacturing companies for their help in overcoming many of the obstacles that were present in making these kits widely available. This is why, with decades of experience in comparable diagnostic test kit manufacturing, Micron was well prepared to rise to the challenge when a global medical equipment producer approached the team seeking the guidance and capabilities that needed to get their COVID-19 home test kit to market.
Reshoring: Reducing Costs and Returning Jobs to The United States
Reshoring is the act of bringing manufacturing jobs that had been previously exported to foreign countries back to the United States. On a national scale, returning jobs to the United States combats high unemployment, reduces trade and budget deficits, and bolsters the economy. Locally, manufacturing companies also benefit from reshoring by being an employment opportunity creator in their respective communities, but most notably benefits by reducing their products’ total cost and improving their balance sheets.
We Is Greater Than Me
We all have expressions we use that are representative of our cultures, our upbringing, our life experiences, or even just something we heard someone say that struck a chord within us. There are the famously inspirational sayings, like Vince Lombardi’s classic quote “Winners never quit and quitters never win," and the often over-used (but still very true), “There is no ‘I’ in ‘team’.” (Peter Drucker)
Applying QMS Fundamentals to Support Organizational Growth
Growth is good—in fact, growth is necessary for survival. But just as the physical growth in humans comes with challenges, so does organizational growth. Unlike human growth, however, there are aspects of an organization’s growth that are within their control. One aspect of organizational growth that must be controlled is the quality of the organization’s processes and products to ensure that quality must never suffer at the expense of growth.
How Automated Manufacturing Improves ROI
Contract manufacturing breeds innovation, but without the right experience to guide process engineering, it can be difficult to break out of established patterns. In this article we will compare the automated processes applied to two similar-yet-different products. While these items perform nearly identical functions, each has its own distinct assembly and packaging requirements. While many contract manufacturers would simply produce the parts simultaneously and add manual operations to each line, the team at Micron uncovered unique opportunities to increase efficiency and ROI by approaching each item as a unique part.
Micron Products is now Micron Solutions
Micron Products is expanding and changing its name to Micron Solutions to reflect an enhanced scope of services, including turnkey, complex assembly.
How to Know When Automated Manufacturing Is Right For You
Industrial technology has progressed at a breakneck pace recently, and the application of robotic technology and automation is a fundamental tool in modern manufacturing. According to the Boston Consulting Group, “combining advanced robotics with other technologies, process enhancements, and structural layout changes [in a manufacturing environment] can yield savings of up to 40%,” which presents considerable potential for manufacturing companies seeking to leverage the expertise of a skilled contract manufacturing partner.
Tooling and Mold-Making at Micron
Mold making has a long history and it is no exception at Micron where we have mold makers with 35 to 45 years of experience under their belts. Our goal is to make quality, well-built molds for various industries and, with our experienced mold makers, we can achieve that.
The key to producing a high-quality mold is precise tolerances, maximum production performance and quality mold design. We are able to create these quality molds with the support of advanced machining equipment, a good quality team and the support of a qualified engineering department.
What Is Quality, Really?
There are almost as many definitions for Quality as there are quality practitioners. Some common ones include:
- Conforming to a quality standard
- Meeting customer specifications
- The standard of something as measured against other things of a similar kind
- Suitability for intended use
But what is Quality, really?
Why Leading Innovators of COVID-19 Home Testing Are Choosing Micron
The first half of 2020 has been, for many, a grueling gauntlet of disruptions to normal work and social behaviors. With the weather warming, people are demanding a quick and clear pathway to reopening schools, businesses, and social activities in a safe, reasonable manner. Public health officials have outlined several steps that must occur to safely resume normal activity, but one of the most prominent requirements consistently emphasized has been the need for widely-available, accurate, easy-to-use home testing for COVID-19. Calling upon years of experience manufacturing various components and consumables for medical testing kits, Micron is now working with some of the medical manufacturers leading the way for COVID-19 testing to become widely available in simple, single-use home test kits.
COVID-19 Notice
The world community faces unprecedented challenges in response to COVID-19, including increasing demand for essential medical devices throughout a period of global supply chain disruption.
Micron attends MEDICA Trade Fair in Dusseldorf Germany
11 Questions To Ask Before Picking A Plastic Mold Manufacturing Partner
When you’re getting ready to begin the injection molding process, the first choice you make—and one of the most crucial decisions—is which plastic mold manufacturing partner you’ll select. The partner you choose should, of course, deliver on all your mold requirements—but they should also prototype your part, help you with part design adjustments, warranty their work, and much more. And most importantly, the right partner will ensure you don’t end up with a useless mold that doesn’t produce quality parts—or, as we like to call a faulty mold—a boat anchor.
Precision Molding: Three Questions That Could Save You Money
If you need a plastic part molded with extreme precision—for example, to ensure there’s no air leak between two molded sections or to be certain there’s no visible seal gap line—you likely require precision molding. The difference between a typical injection molded part and a precision molded part is the tolerance, or acceptable range of variation in dimension: While the majority of injection molded parts have a tolerance of +/- .005″, precision molding holds tolerances between +/- .002″ and +/- .001″ (or less, in some cases).
Manufacturing Single-Use Products: 7 Things To Keep In Mind
A single-use product is meant to be used or applied once and then discarded. The term “single-use” is sometimes intended to mean “disposable,” though this isn’t always the case. A bullet and a booster rocket, for example, are both single-use products—but most people wouldn’t refer to them as a disposable. But many medical products, like tongue depressors and test vials, are perfect examples of single-use, disposable products.
3 Common Types Of Rapid Tooling Used In Prototyping
Rapid tooling is, simply, the creation of a mold in a shortened timeline.
Rapid tooling got its start in the 1990s, when engineers involved in injection molding wanted to see if they could build molds in a matter of hour or days instead of the weeks or months a machined mold would take. A rapid-tooled mold is ideal for prototyping a part and molding a few hundred plastic parts before full-scale, high-volume production starts.
Blow Molding Vs. Injection Molding: A Comparison For Engineers
Blow molding vs. injection molding—what’s the difference? Both are common methods used to create plastic parts. And while some parts require both blow-molded and injection-molded components—for example, a medical device with a blow-molded container attached to an injection molded apparatus, or a military application with a blow-molded “payload” packet fabricated inside an injection molded projectile—the two methods primarily serve different markets.
What Type Of Molding Process Does Your Prototype Require?
If you’re creating a plastic part, it’s important to know what type of injection molding process your part will require. Do you know if your part needs to be molded in a cleanroom environment, or whether you should use a vertical or horizontal injection molding machine?
Plastic Mold Manufacturing: A Behind-The-Scenes Look At Micron
There are thousands of plastic injection molding companies around the world, but here at Micron, we like to think we approach things a little differently from the rest of the crowd.
One of those differences is that we do our plastic mold manufacturing in-house as opposed to outsourcing this job. This allows us a high degree of quality control throughout the mold-making process, and helps to address any questions or issues on an injection molding project before the mold is created.
Cleanroom Molding: What You Need To Know Before Manufacturing
If you need to manufacture a plastic part that must remain as clean as possible—like an implantable medical device—you'll need to have your part made in a cleanroom environment.
Cleanroom molding is the process of creating plastic parts in a special room optimized to reduce the risk of contamination by dust or other particles. The medical, pharmaceutical, aerospace, military, and biotech industries frequently require parts to be created in a cleanroom environment.
The Use Of 3D Printing In Manufacturing Now & In The Future
In recent years, 3D printing has become extremely useful in manufacturing—and, more specifically, in plastic injection molding. Injection molding companies often use a 3D printer to create a part from a model, drawing, or concept plastic part.
6 Types Of Injection Molding Technology
From advancements that have helped the industry for over 40 years to the latest cutting-edge innovations, there are a number of interesting plastic injection technologies out there that could be used to bring your prototype into production.
Meet Micron: An Innovative Injection Molding Company In Massachusetts
Hello! We’re Micron Products.
Founded in 1972, we are a full-service contract manufacturing and injection molding company based in Massachusetts. The shop we acquired at that time was founded in 1925, making us one of the oldest companies in the injection molding industry.
How Does The Injection Molding Process Work? A Breakdown For Product Engineers
As an engineer, your focus is on taking a product idea and figuring out how to get it manufactured so it fits all your specifications and stays within your budget. But before you select an injection molding partner, it’s a good idea to brush up on what the injection molding process looks like.
Mold Making Services In The U.S. Vs. China—Which Should You Choose?
If your company requires plastic injection molding services to create a plastic part, one of the first—and most critical—decisions to make is whether you’re going to go with a plastic injection mold service in the U.S. or one based overseas. While many countries offer mold-making services, China is the primary player in this market.
Medical Device Injection Molding: How To Find The Right Partner
Medical device injection molding is used in everything from syringes to IV roller clamps to dialysis machine components.
While you must ensure that your medical device is manufactured to FDA standards and is ISO 13485 compliant, you also need to be certain that the company you select is the right one for your needs.
Thermoset Vs. Thermoplastic Injection Molding: A Comparison
The properties of thermosets and thermoplastics are quite different—but the similarities and differences are often asked about. The primary difference in these two plastics comes down to heat and chemical resistance. Heat resistance is the primary function of a thermoset material, while thermoplastics—which are much more common—can only withstand heat to a certain degree. It’s worth noting that plastic injection molding companies typically do either thermoset or thermoplastic injection molding, but rarely do they handle both.
Injection Molding Terms Every Engineer Should Know
The plastic injection molding process is extremely complex with (quite literally) thousands of moving parts. As a manufacturing engineer, it’s not critical for you to know every finite detail of mold-closing mechanisms or the difference between every polymeric substance used in injection molding—but understanding the following 10 terms will make a conversation with a potential plastic manufacturing partner much simpler.